Wink of Knowledge: Density measurement during inerting
During inerting, an inert gas such as nitrogen or CO2 protects against explosions, corrosion, and other harmful effects. The process displaces oxygen or hazardous substances from the system. Only when their concentration falls below the relevant safety threshold—e.g., below 4% for hydrogen—is the system considered inerted and can be maintained or operated safely.
Why this test?
In classic applications, inerting is usually understood as displacing oxygen to prevent reactions or explosions. Accordingly, dedicated oxygen sensors are well established. Equally important, however, is displacing flammable, harmful, or explosive gases when a system must be ventilated—and unfortunately oxygen sensors provide no help in monitoring the hazardous substances themselves. A very universally applicable measurand would be density measurement for determining concentration. This value would also complement the existing O2 measurement: the density of O2 is too close to N2 to be used sensibly, but the density difference to flammable substances such as H2, methane, or LPG is very large. In this “knowledge wink,” we look at an example purge process with N2 and use H2 as a representative hazardous substance.
What is a Wink of Knowledge?
Do you need to quickly measure, draw or do/build something? The speed with which the result may be achieved counts more than the perfect (scientific) approach. For this reason, we have introduced the Wink of Knowledge. Science in the wink of an eye, so to speak. We don’t want to prove anything scientifically. We simply want to quickly demonstrate something pragmatically. If you are interested, we would be happy to discuss these results in more detail with you and your project.
Results
To avoid hazardous substances, we use helium as a surrogate for hydrogen. After hydrogen, helium has the second-lowest density of all pure gases and is therefore the best possible alternative. Under ambient conditions, H2 is approx. 0.08 kg/m3 and He approx. 0.16 kg/m3. All other gases have a significantly higher density; the most commonly used inert gas N2, for example, has a density of approx. 1.14 kg/m3, and CO2 even 1.81 kg/m3.
In the test setup, we use a DGF-I1, which we calibrated to the two pure gases N2 and He. It was installed in a pipe section that is open to ambient air. This roughly corresponds to the installation situation at the outlet of a gas tank or an electrolyzer plant. It should be noted that atmospheric pressure is more challenging for the density sensor than overpressure. Increasing the pressure reduces the relative measurement error and thus the noise in the concentration measurement. The test therefore represents the “worst case” in terms of precision.
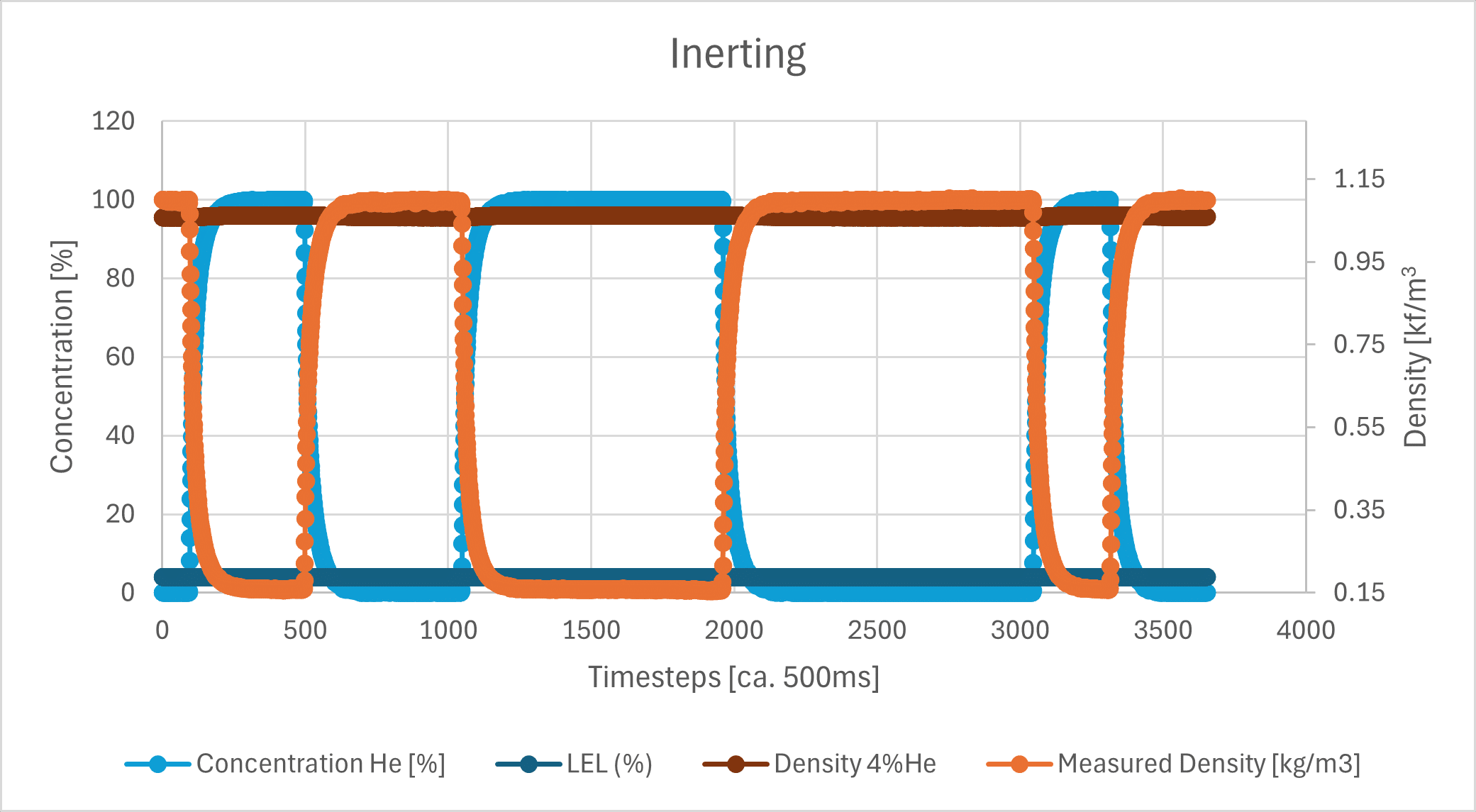
However, the sensor responded very quickly to the applied gas changes between He and N2 and reliably reached the expected end values between 100% He and 100% N2. The curves show the typical asymptotic dilution behavior of a purge process:
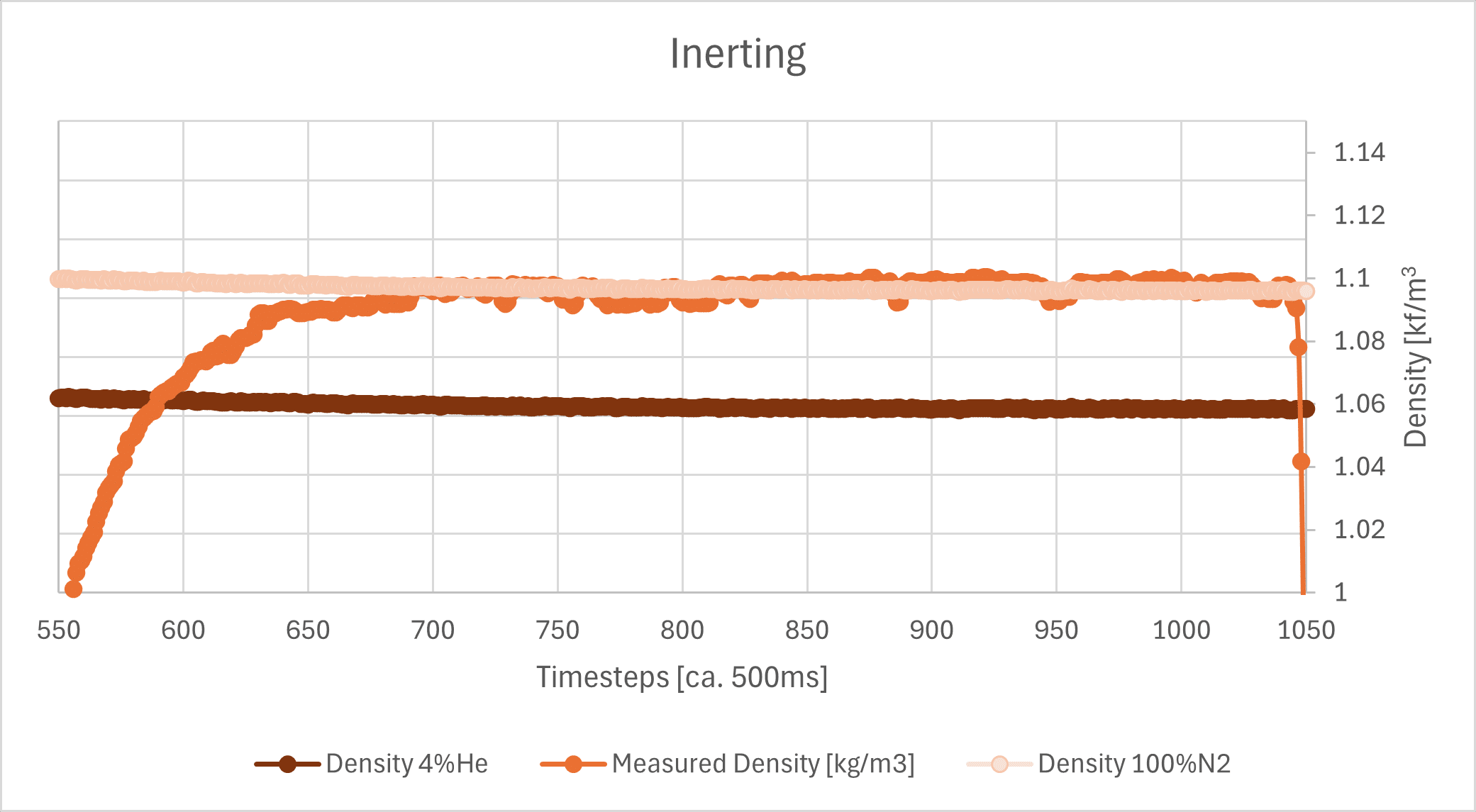
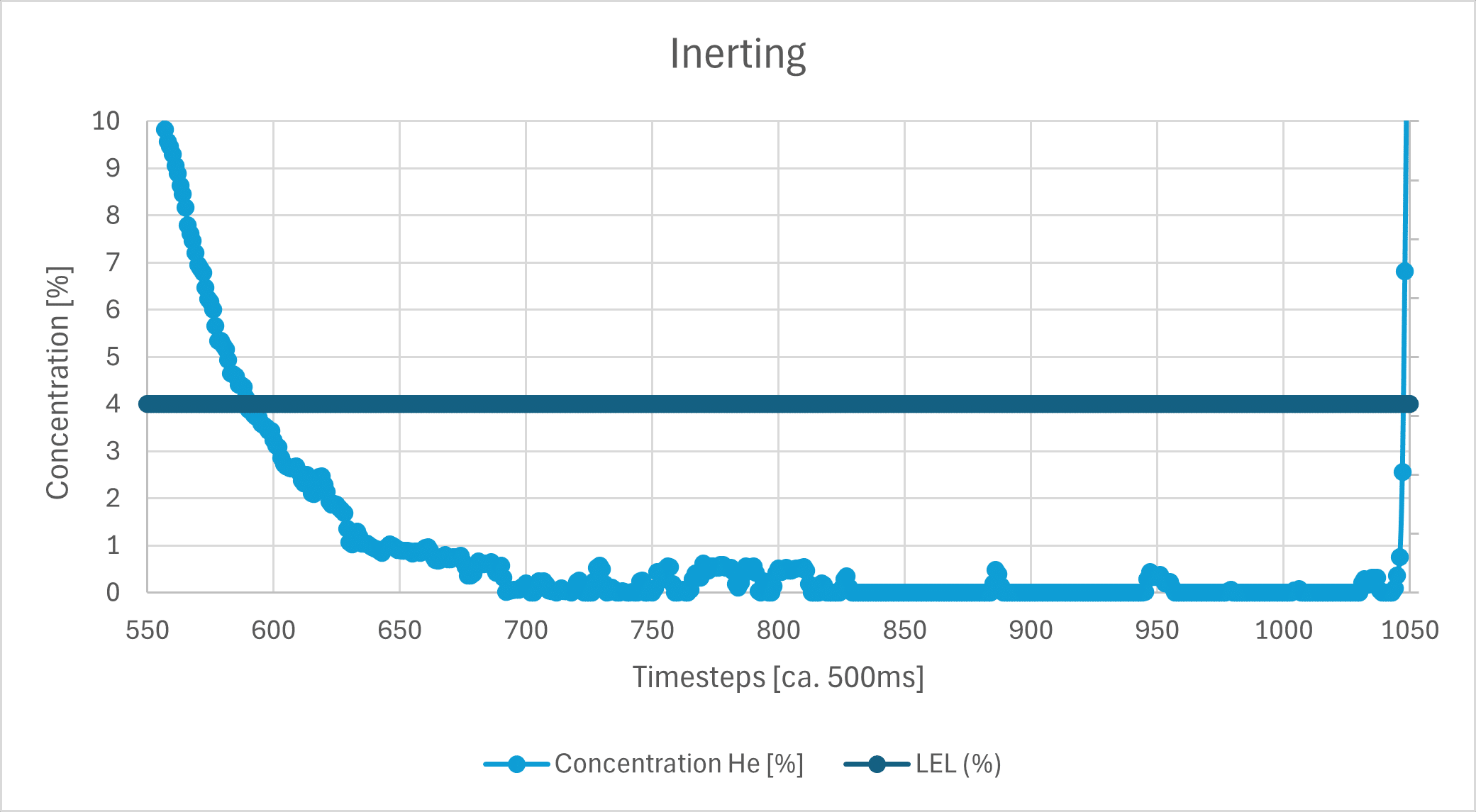
Also shown is the hypothetical explosive limit for H2, which is 4% in air (dark blue line), as well as the calculated density value at a He concentration of 4% (dark brown line). Since these values are very close to the extreme values, here is a zoom-in:
Even the unfiltered sensor noise at approx. a 500 ms measurement interval is clearly lower than the typically required accuracy of 1%. Further signal processing, such as a moving average, can improve the signal even more.
The measurement campaign thus clearly demonstrates that density measurement can be used for precise concentration measurement of hazardous substances during inerting. In addition to the example shown here, the measurand can be used for a wide range of gas combinations. Feel free to contact us with your inerting task!
Procedure
Two Vögtlin mass flow controllers were used to generate a constant flow rate of 0.5 nl/min (helium and nitrogen alternately). The DGF was connected as follows:
The pipe section was open to the surroundings at all times, and the pressure was therefore approximately 1 bar. The DGF-I1 was configured and read out using the free Remote Control software. Before the measurement series started, the sensor was calibrated to the pure gases. The values read out were temperature, pressure, density, and concentration (N2/He mixture) at a nominal rate of 300 ms (in reality approx. 500 ms). Using the pressure and temperature values, the density for 100% N2 and for 96% N2 / 4% He was calculated with NIST REFPROP.
Conclusion
We demonstrated the use of density measurement for monitoring inerting. Inert gases such as nitrogen or CO2 are used to displace oxygen and flammable gases in order to prevent explosions and corrosion.
- Inerting protects against hazards: Inert gases displace oxygen or flammable gases, so the system is only considered inert once safety-relevant concentration thresholds are undershot, e.g., below 4% for hydrogen.
- Density measurement as a universal measurand: In addition to oxygen sensors, density measurement enables concentration determination of hazardous substances because the density differences between inert gases and flammable gases such as hydrogen or methane are large.
- Experimental proof with helium: Helium serves as a hydrogen substitute due to its similarly low density. In a purge process with helium and nitrogen, a DGF-I1 sensor showed fast, precise responses to gas switching at ambient pressure, even close to the explosive limit of hydrogen.
- Precision and application: The sensor signal shows low noise and meets accuracy requirements of below 1%. Density measurement is therefore well suited for concentration monitoring of hazardous substances during inerting.
Sensors that might interest you
Gases
Viscosity
Applications that might interest you

From volume (l)
to mass (kg)
More Winks of Knowledge that might interest you
Article: In-line measurements of the physical and thermodynamic properties of single and multicomponent liquids
Microfluidic devices are becoming increasingly important in various fields of pharmacy, flow chemistry and healthcare. In the embedded microchannel, the flow rates, the dynamic viscosity of the transported liquids and the fluid dynamic properties play an important role. Various functional auxiliary components of microfluidic devices such as flow restrictors, valves and flow meters need to be characterised with liquids used in several microfluidic applications.
Article: Density and Concentration Measurement Applications for Novel MEMS-based Micro Densitometer for Gas
Density and Concentration Measurement Applications for Novel MEMS-based Micro Densitometer for GasC. Huber, TrueDyne Sensors AG, Reinach BL (Switzerland), Endress+Hauser Flowtec, Reinach BL (Switzerland)Abstract A MEMS cantilever based resonant device for gas...
Article: Design, Simulation, Fabrication and Characterization of piezoelectric MEMS Cantilever for Gas Density and Viscosity Sensors Applications
Design, Simulation, Fabrication and Characterization of piezoelectric MEMS Cantilever for Gas Density and Viscosity Sensors ApplicationsA. Mehdaoui¹, C. Huber¹, J. Becker¹, F. Schraner¹, L. Villanueva² ¹TrueDyne Sensors AG, Reinach BL (Switzerland), ²Ecole...
Article: Multiparameter Gas-Monitoring System
The aim of the study is to develop a compact, robust and maintenance free gas concentration and humidity monitoring system for industrial use in the field of inert process gases. Our multiparameter gas-monitoring system prototype allows the simultaneous measurement of the fluid physical properties (density, viscosity) and water vapor content (at ppm level) under varying process conditions.
Part 3 – MEMS technology
MEMS technologyAt a glance In the previous section (part 2) we got to know the vibration measuring method. This section deals with the establishment of MEMS technology at TrueDyne Sensors AG. The technology has brought about the MEMS sensor, the heart of which is an...
Part 2 – The resonator density measurement
The resonator density measurement At a glance In the previous part (1) we learned about the basics of density measurement and the definition of density. This section is dedicated to the vibration method which is also used by density sensors for density measurement....